
Taking Patients With Atrial Fibrillation on a VIRTUAL-SAFARI With WatchPAT® ONE At-Home Sleep Apnea Testing
As many as 3 in 4 patients with atrial fibrillation (AFib) have obstructive sleep apnea.1 While treatments such as continuous
Undiagnosed sleep apnea is a major cardiovascular disease risk factor, making the link between sleep apnea and cardiac health a hot topic in healthcare. Read ZOLL Itamar Cardio Sleep Blog for the latest industry news and medical research related to sleep apnea diagnosis and treatment.

As many as 3 in 4 patients with atrial fibrillation (AFib) have obstructive sleep apnea.1 While treatments such as continuous

2021 AHA Recommendations on Obstructive Sleep apnea The American Heart Association released a new Scientific Statement on Obstructive Sleep Apnea
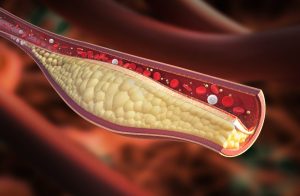
Several groups have reported qualitative associations between obstructive sleep apnea (OSA) and coronary plaque1-3; however, a recent study published in
A recent article in Medical News Today reported on the connection among obstructive sleep apnea (OSA), systemic hypertension (SH), and

Multiple reports show that obstructive sleep apnea reduces the efficacy of catheter ablation treatment for atrial fibrillation.1-5 Indeed, severe obstructive

A post hoc analysis of the Randomized Intervention with CPAP in Coronary Artery Disease and Obstructive Sleep Apnea (RICCADSA) trial

On-pump vs. off-pump coronary artery bypass grafting (CABG) Off-pump CABG avoids complications that can arise with the cardiopulmonary bypass and

Central sleep apnea is common among patients with chronic heart failure with reduced left ventricular ejection fraction (HFrEF), affecting about

Sleep-disordered breathing and heart failure appear to have a bidirectional relationship.1 Obstructive sleep apnea is an independent risk factor to

Gestational diabetes can have short- and long-term consequences for the health of the mother and the fetus. Mothers are more

CPAP Reduces Long-Term Cardiovascular Risk in Patients with Obstructive Sleep Apnea Multiple lines of research show patients with moderate to
Optimal management of OSA may be considered to reduce AF incidence, progression, recurrences, and symptoms. Screening for AF should be